
SOLVED: The alcohol content of vodka is normally given in "proof", which is defined as twice the percentage by volume of ethanol (C2H5OH) present. Calculate the proof of a sample of vodka
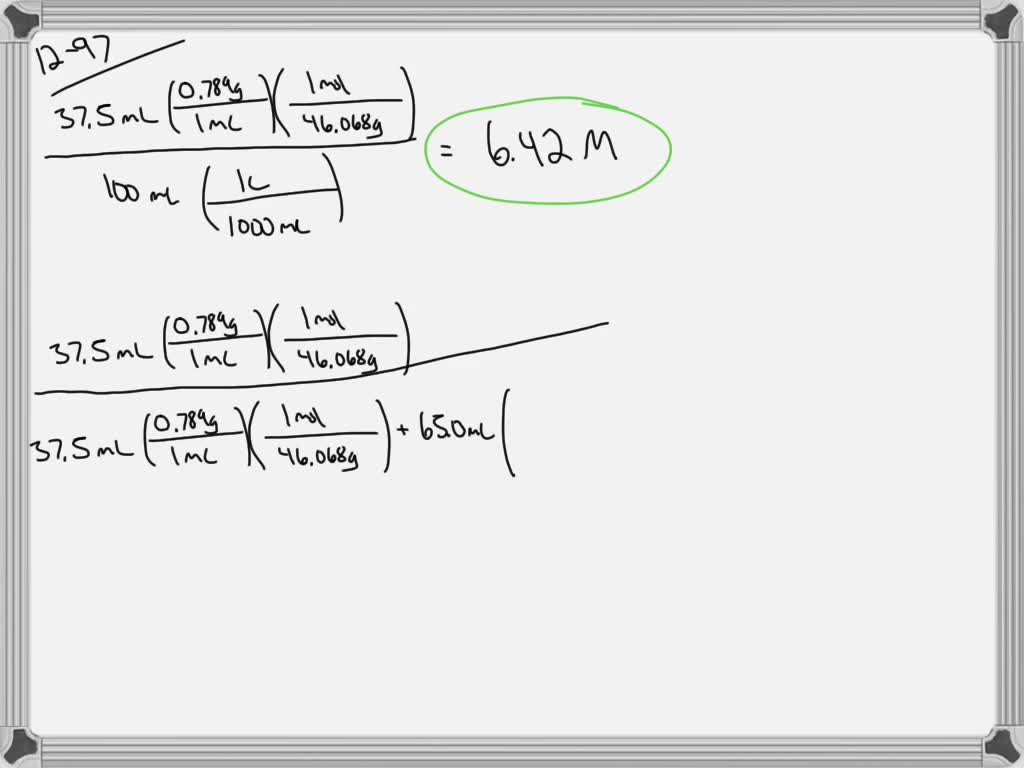
SOLVED:Vodka from the European Union must be at least 37.5 % alcohol by volume or 75 proof. If the density of water is 1.00 g mL^-1 and the density of ethanol is

Structurability: A Collective Measure of the Structural Differences in Vodkas | Journal of Agricultural and Food Chemistry
Densities of vodka brands mapped on the density profile measured for... | Download Scientific Diagram
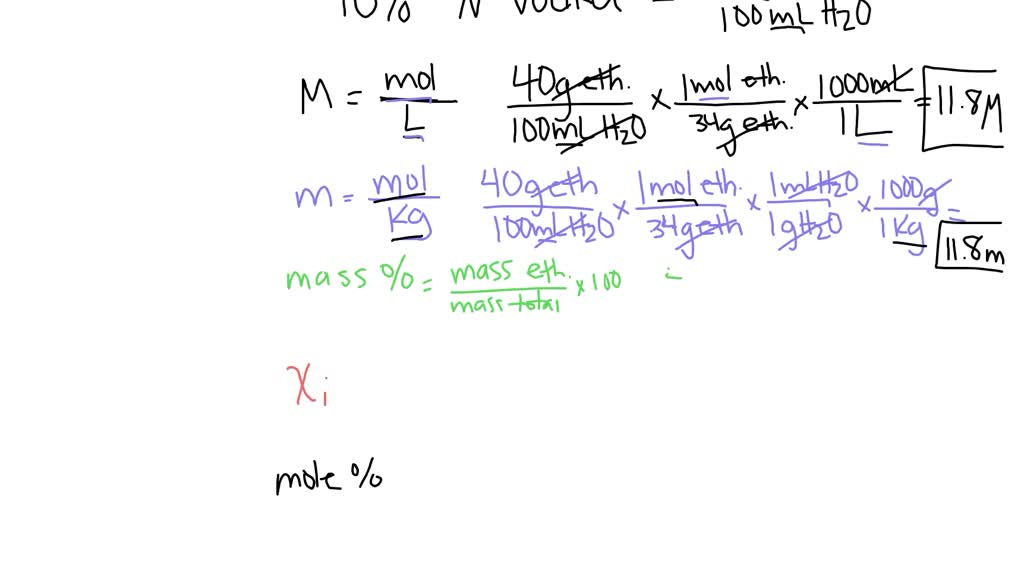
SOLVED: Calculate the concentration of vodka (40.0%, by volume, ethanol CH3CH2OH in water) in the following units. Assume the density of the solution is 0.942 g/mL, pure water is 1.00 g/mL, and

Density Chart for 2 #LayerDrinks: We have tried our best to determine the exact density of the most popular liquors, but sho… | Smirnoff vodka, Distillation, Liquor

SOLVED: 1. Vodka is advertised to be 80 proof, which is 40% by volume of ethanol, C2H5OH. Assuming the density of the solution is 1.0 g/mL and the density of ethanol is

SOLVED: Chem 121 24. If vodka contains 37.0% alcohol by volume, how many mL of alcohol are present in one bottle of vodka that weighs 557 g? (Density of vodka is 1.07
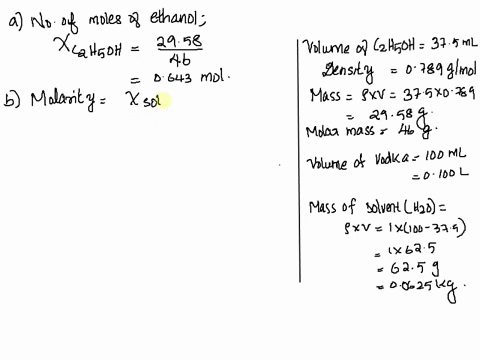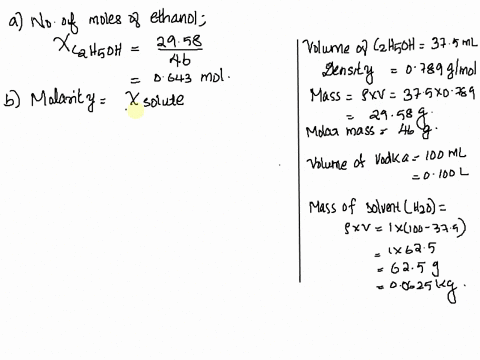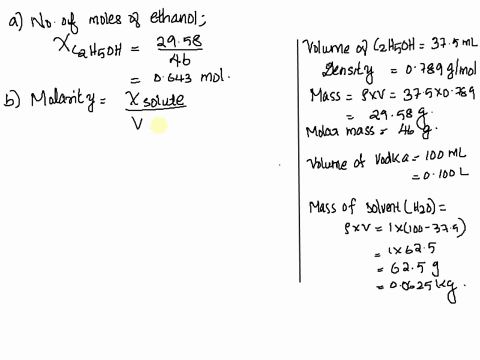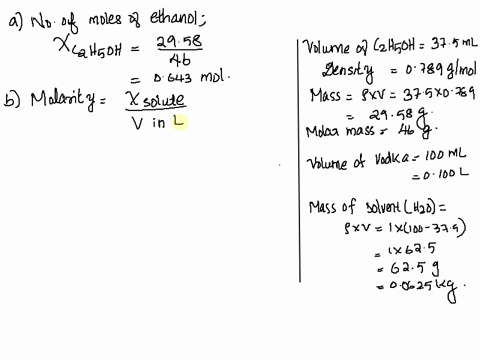
SOLVED: Vodka sold in Europe is a 37.5% aqueous solution of ethanol (C2H6O). The density of vodka is 0.916 g/mL. What is the mole fraction, molality, and molarity of ethanol in vodka?

wooden box alcoholmeter for Vodka/whiskey alcohol meter wine alcohol tester Alcohol hydrometer density meter - AliExpress

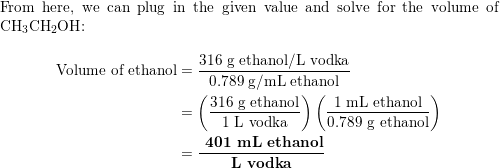













:max_bytes(150000):strip_icc()/_irish-flag-shooter-recipe-759704-04-5bd71afec9e77c00581eae40.jpg)
